| Tag | Content | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
SG ID |
SG00000116 |
|||||||||||||||||||||
UniProt Accession |
||||||||||||||||||||||
Theoretical PI |
6.06
|
|||||||||||||||||||||
Molecular Weight |
317245 Da
|
|||||||||||||||||||||
Genbank Nucleotide ID |
||||||||||||||||||||||
Genbank Protein ID |
||||||||||||||||||||||
Gene Name |
brca2 |
|||||||||||||||||||||
Gene Synonyms/Alias |
||||||||||||||||||||||
Protein Name |
||||||||||||||||||||||
Protein Synonyms/Alias |
SubName: Uncharacterized protein |
|||||||||||||||||||||
Organism |
Danio rerio (Zebrafish) (Brachydanio rerio) |
|||||||||||||||||||||
NCBI Taxonomy ID |
7955 |
|||||||||||||||||||||
Chromosome Location |
|
|||||||||||||||||||||
Function in Stage |
||||||||||||||||||||||
Function in Cell Type |
||||||||||||||||||||||
Description |
Temporarily unavailable |
|||||||||||||||||||||
The information of related literatures |
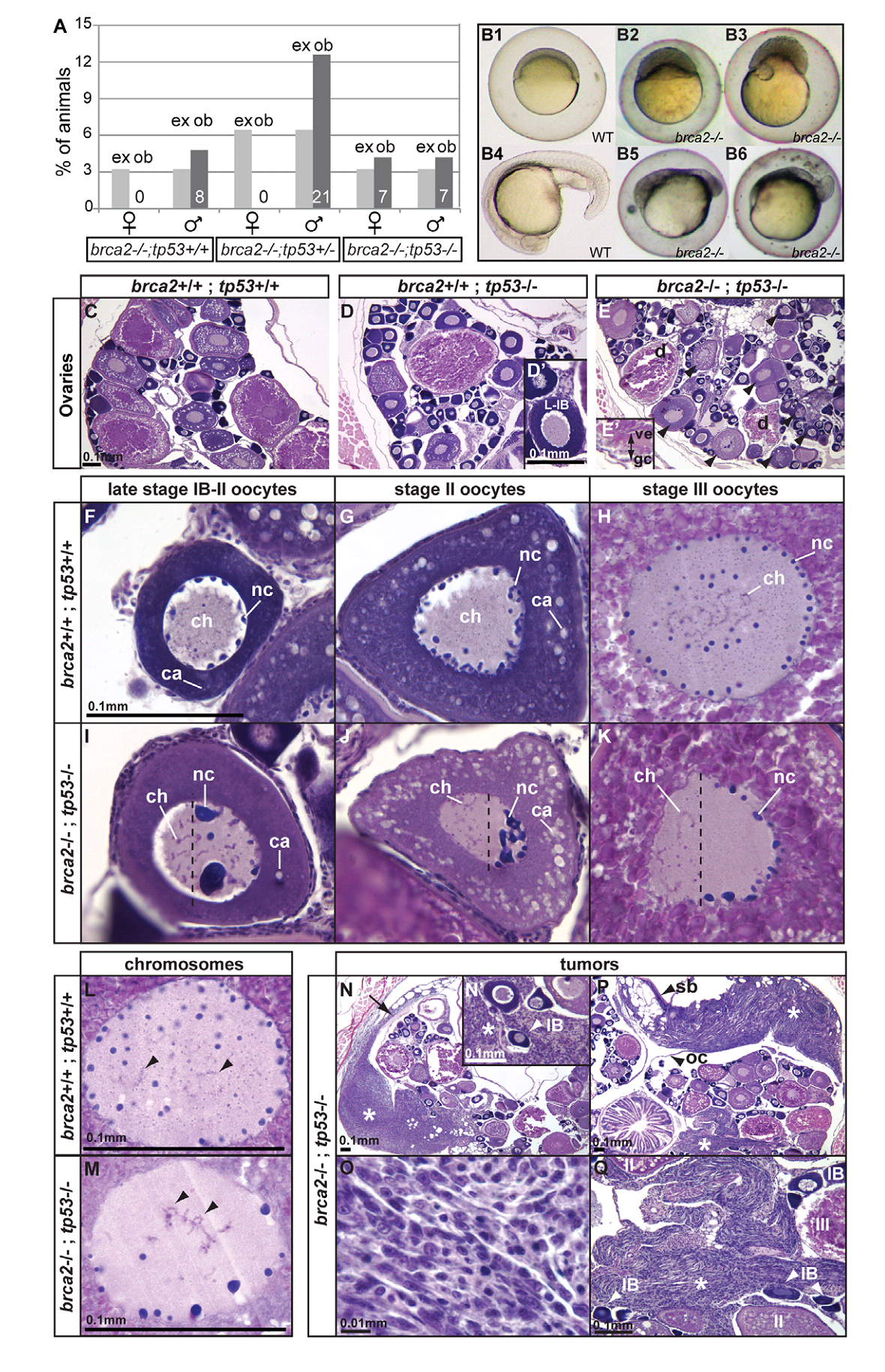
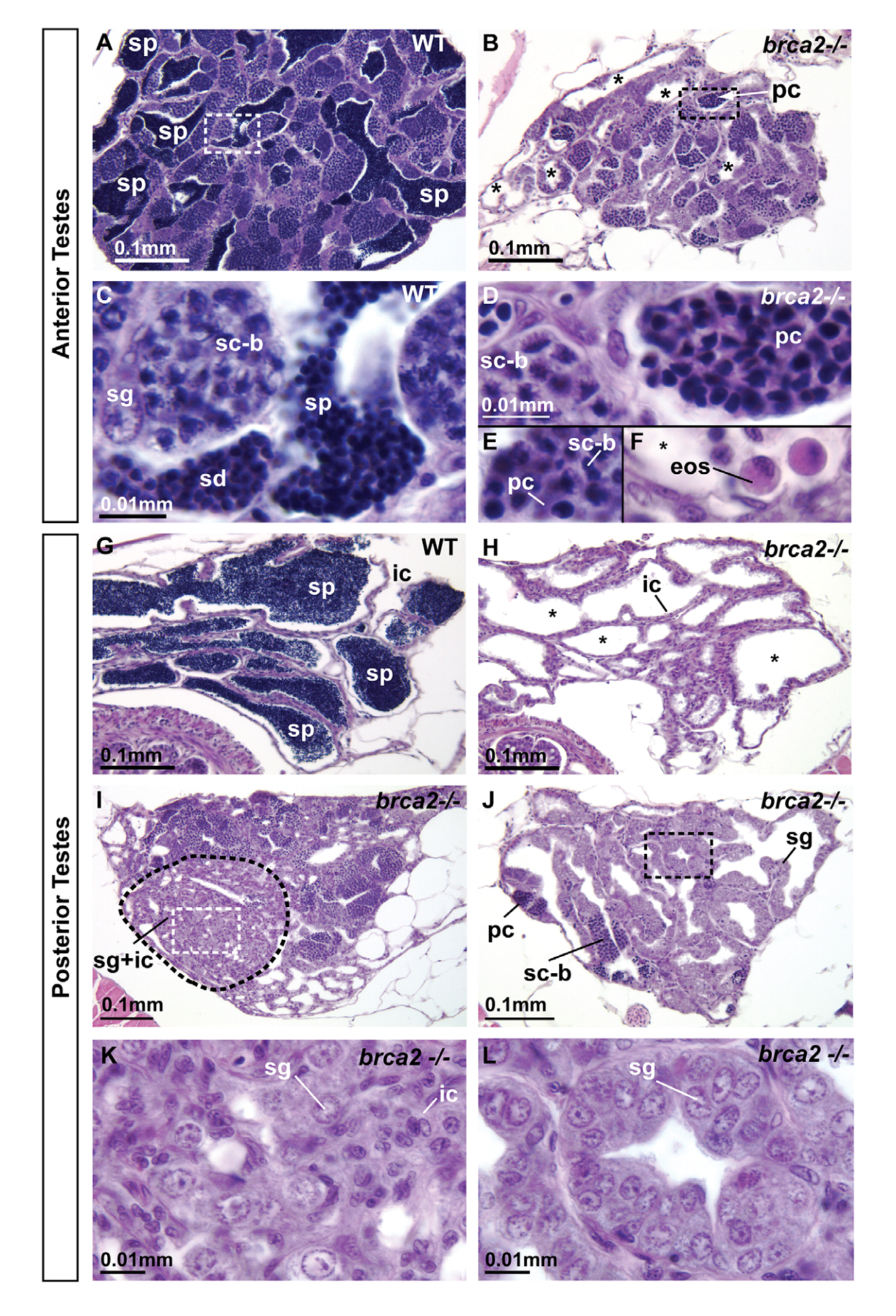
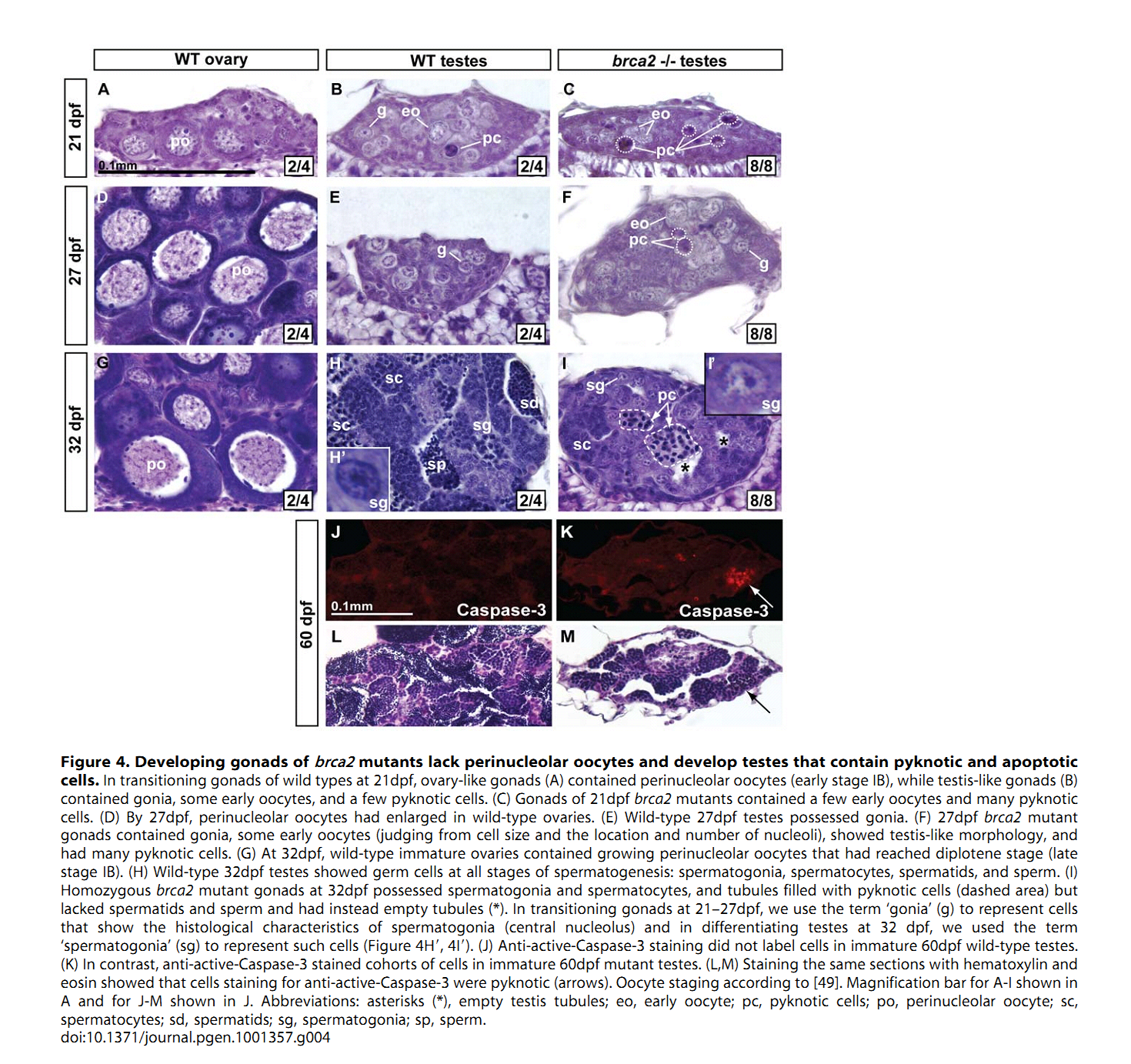
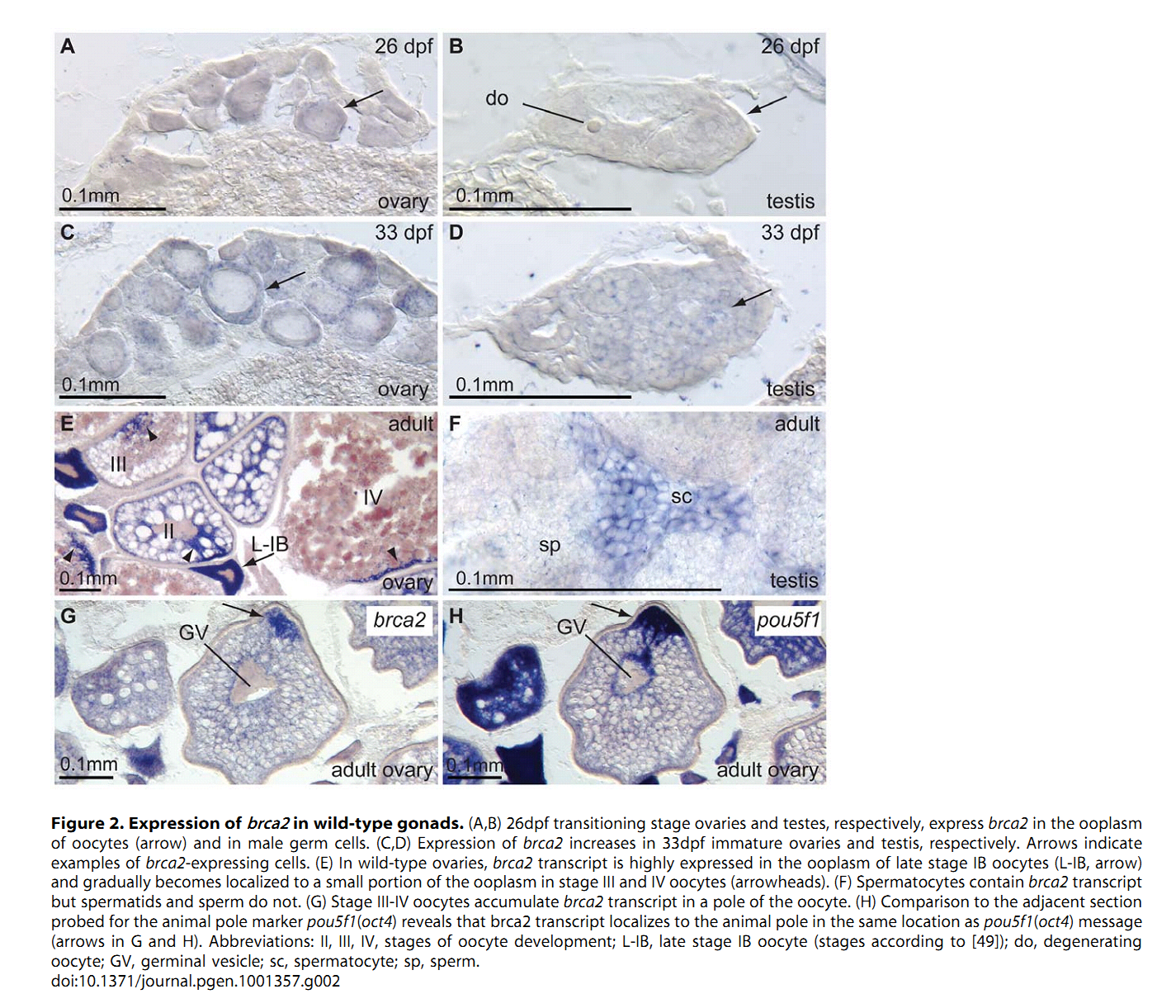
1. A. Rodriguez-Mari, C. Wilson, T. A. Titus, C. Canestro, R. A. BreMiller, Y. L. Yan, I. Nanda, A. Johnston, J. P. Kanki, E. M. Gray, X. He, J. Spitsbergen, D. Schindler and J. H. Postlethwait (2011) Roles of brca2 (fancd1) in oocyte nuclear architecture, gametogenesis, gonad tumors, and genome stability in zebrafish. PLoS Genet 7(3): e1001357. Abstract Mild mutations in BRCA2 (FANCD1) cause Fanconi anemia (FA) when homozygous, while severe mutations cause common cancers including breast, ovarian, and prostate cancers when heterozygous. Here we report a zebrafish brca2 insertional mutant that shares phenotypes with human patients and identifies a novel brca2 function in oogenesis. Experiments showed that mutant embryos and mutant cells in culture experienced genome instability, as do cells in FA patients. In wild-type zebrafish, meiotic cells expressed brca2; and, unexpectedly, transcripts in oocytes localized asymmetrically to the animal pole. In juvenile brca2 mutants, oocytes failed to progress through meiosis, leading to female-to-male sex reversal. Adult mutants became sterile males due to the meiotic arrest of spermatocytes, which then died by apoptosis, followed by neoplastic proliferation of gonad somatic cells that was similar to neoplasia observed in ageing dead end (dnd)-knockdown males, which lack germ cells. The construction of animals doubly mutant for brca2 and the apoptotic gene tp53 (p53) rescued brca2-dependent sex reversal. Double mutants developed oocytes and became sterile females that produced only aberrant embryos and showed elevated risk for invasive ovarian tumors. Oocytes in double-mutant females showed normal localization of brca2 and pou5f1 transcripts to the animal pole and vasa transcripts to the vegetal pole, but had a polarized rather than symmetrical nucleus with the distribution of nucleoli and chromosomes to opposite nuclear poles; this result revealed a novel role for Brca2 in establishing or maintaining oocyte nuclear architecture. Mutating tp53 did not rescue the infertility phenotype in brca2 mutant males, suggesting that brca2 plays an essential role in zebrafish spermatogenesis. Overall, this work verified zebrafish as a model for the role of Brca2 in human disease and uncovered a novel function of Brca2 in vertebrate oocyte nuclear architecture. PMID: [21483806] 2. H. R. Shive, R. R. West, L. J. Embree, M. Azuma, R. Sood, P. Liu and D. D. Hickstein (2010) brca2 in zebrafish ovarian development, spermatogenesis, and tumorigenesis. Proc Natl Acad Sci U S A 107(45): 19350-5. Abstract Humans with inherited mutations in BRCA2 are at increased risk for developing breast and ovarian cancer; however, the relationship between BRCA2 mutation and these cancers is not understood. Studies of Brca2 mutation by gene targeting in mice are limited, given that homozygous Brca2 mutation typically leads to early embryonic lethality. We established a zebrafish line with a nonsense mutation in brca2 exon 11 (brca2(Q658X)), a mutation similar in location and type to BRCA2 mutations found in humans with hereditary breast and ovarian cancer. brca2(Q658X) homozygous zebrafish are viable and survive to adulthood; however, juvenile homozygotes fail to develop ovaries during sexual differentiation. Instead, brca2(Q658X) homozygotes develop as infertile males with meiotic arrest in spermatocytes. Germ cell migration to the embryonic gonadal ridge is unimpaired in brca2(Q658X) homozygotes; thus, failure of ovarian development is not due to defects in early establishment of the embryonic gonad. Homozygous tp53 mutation rescues ovarian development in brca2(Q658X) homozygous zebrafish, reflecting the importance of germ cell apoptosis in gonad morphogenesis. Adult brca2(Q658X) homozygous zebrafish are predisposed to testicular neoplasias. In addition, tumorigenesis in multiple tissues is significantly accelerated in combination with homozygous tp53 mutation in both brca2(Q658X) homozygous and brca2(Q658X) heterozygous zebrafish. These studies reveal critical roles for brca2 in ovarian development and tumorigenesis in reproductive tissues. PMID: [20974951] Back to Top |
|||||||||||||||||||||
Figures for illustrating the function of this protein/gene |
|
|||||||||||||||||||||
Function |
||||||||||||||||||||||
Subcellular Location |
||||||||||||||||||||||
Tissue Specificity |
||||||||||||||||||||||
Gene Ontology |
|
|||||||||||||||||||||
Interpro |
||||||||||||||||||||||
Pfam |
||||||||||||||||||||||
SMART |
||||||||||||||||||||||
PROSITE |
||||||||||||||||||||||
PRINTS |
||||||||||||||||||||||
Created Date |
18-Oct-2012 |
|||||||||||||||||||||
Record Type |
Experiment identified |
|||||||||||||||||||||
Protein sequence Annotation |
||||||||||||||||||||||
Nucleotide Sequence |
Length: bp Go to nucleotide: FASTA |
|||||||||||||||||||||
Protein Sequence |
Length: 2874 bp Go to amino acid: FASTA |
|||||||||||||||||||||
The verified Protein-Protein interaction information |
| |||||||||||||||||||||
Other Protein-Protein interaction resources |
String database |
|||||||||||||||||||||
View Microarray data |
Temporarily unavailable |
|||||||||||||||||||||
Comments |
||||||||||||||||||||||