| Tag | Content | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
SG ID |
SG00000338 |
|||||||||||||||||||||||||||||||||
UniProt Accession |
||||||||||||||||||||||||||||||||||
Theoretical PI |
9.45
|
|||||||||||||||||||||||||||||||||
Molecular Weight |
11369 Da
|
|||||||||||||||||||||||||||||||||
Genbank Nucleotide ID |
||||||||||||||||||||||||||||||||||
Genbank Protein ID |
||||||||||||||||||||||||||||||||||
Gene Name |
Cyt-c-d |
|||||||||||||||||||||||||||||||||
Gene Synonyms/Alias |
CytC1, DC3 |
|||||||||||||||||||||||||||||||||
Protein Name |
Cytochrome c-1 |
|||||||||||||||||||||||||||||||||
Protein Synonyms/Alias |
Cytochrome c-distal; |
|||||||||||||||||||||||||||||||||
Organism |
Drosophila melanogaster (Fruit fly) |
|||||||||||||||||||||||||||||||||
NCBI Taxonomy ID |
7227 |
|||||||||||||||||||||||||||||||||
Chromosome Location |
|
|||||||||||||||||||||||||||||||||
Function in Stage |
||||||||||||||||||||||||||||||||||
Function in Cell Type |
||||||||||||||||||||||||||||||||||
Description |
Temporarily unavailable |
|||||||||||||||||||||||||||||||||
The information of related literatures |
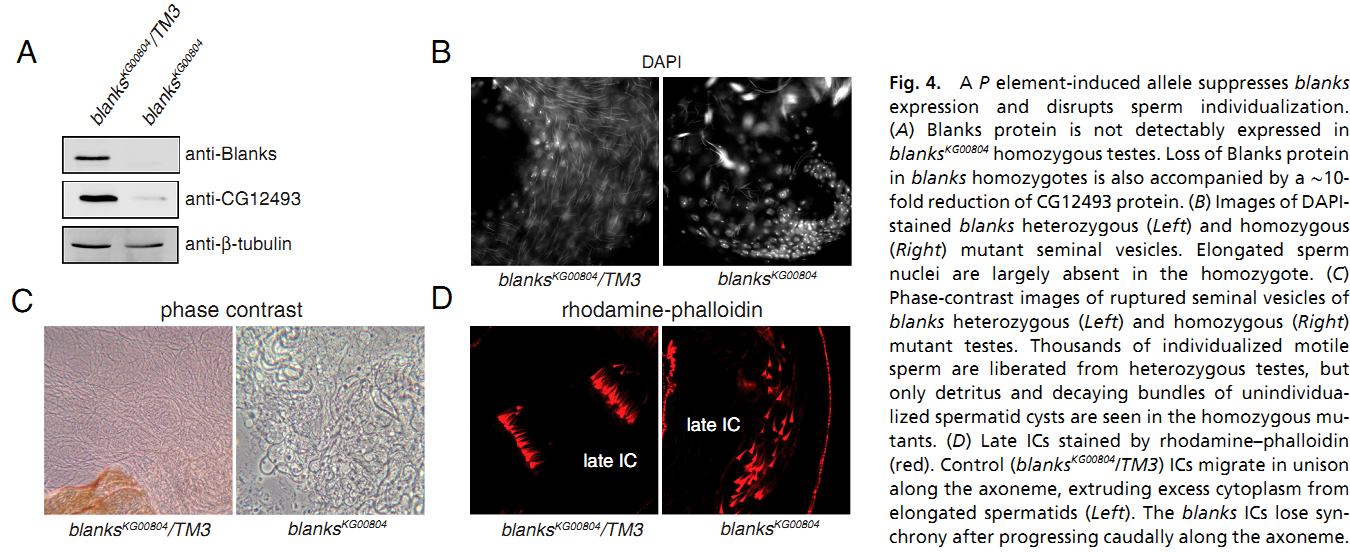
1. V. R. Gerbasi, J. B. Preall, D. E. Golden, D. W. Powell, T. D. Cummins and E. J. Sontheimer (2011) Blanks, a nuclear siRNA/dsRNA-binding complex component, is required for Drosophila spermiogenesis. Proc Natl Acad Sci U S A 108(8): 3204-9. Abstract Small RNAs and a diverse array of protein partners control gene expression in eukaryotes through a variety of mechanisms. By combining siRNA affinity chromatography and mass spectrometry, we have identified the double-stranded RNA-binding domain protein Blanks to be an siRNA- and dsRNA-binding protein from Drosophila S2 cells. We find that Blanks is a nuclear factor that contributes to the efficiency of RNAi. Biochemical fractionation of a Blanks-containing complex shows that the Blanks complex is unlike previously described RNA-induced silencing complexes and associates with the DEAD-box helicase RM62, a protein previously implicated in RNA silencing. In flies, Blanks is highly expressed in testes tissues and is necessary for postmeiotic spermiogenesis, but loss of Blanks is not accompanied by detectable transposon derepression. Instead, genes related to innate immunity pathways are up-regulated in blanks mutant testes. These results reveal Blanks to be a unique component of a nuclear siRNA/dsRNA-binding complex that contributes to essential RNA silencing-related pathways in the male germ line. PMID: [21300896] 2. E. Arama, M. Bader, M. Srivastava, A. Bergmann and H. Steller (2006) The two Drosophila cytochrome C proteins can function in both respiration and caspase activation. EMBO J 25(1): 232-43. Abstract Cytochrome C has two apparently separable cellular functions PMID: [16362035] Back to Top |
|||||||||||||||||||||||||||||||||
Figures for illustrating the function of this protein/gene |
|
|||||||||||||||||||||||||||||||||
Function |
Electron carrier protein. The oxidized form of thecytochrome c heme group can accept an electron from the heme groupof the cytochrome c1 subunit of cytochrome reductase. Cytochrome cthen transfers this electron to the cytochrome oxidase complex,the final protein carrier in the mitochondrial electron-transportchain. Back to Top |
|||||||||||||||||||||||||||||||||
Subcellular Location |
Mitochondrion intermembrane space.Note=Loosely associated with the inner membrane. |
|||||||||||||||||||||||||||||||||
Tissue Specificity |
||||||||||||||||||||||||||||||||||
Gene Ontology |
|
|||||||||||||||||||||||||||||||||
Interpro |
||||||||||||||||||||||||||||||||||
Pfam |
||||||||||||||||||||||||||||||||||
SMART |
||||||||||||||||||||||||||||||||||
PROSITE |
||||||||||||||||||||||||||||||||||
PRINTS |
||||||||||||||||||||||||||||||||||
Created Date |
18-Oct-2012 |
|||||||||||||||||||||||||||||||||
Record Type |
Experiment identified |
|||||||||||||||||||||||||||||||||
Protein sequence Annotation |
INIT_MET 1 1 Removed (By similarity). CHAIN 2 105 Cytochrome c-1. /FTId=PRO_0000108259. METAL 21 21 Iron (heme axial ligand). METAL 83 83 Iron (heme axial ligand). BINDING 17 17 Heme (covalent). BINDING 20 20 Heme (covalent). Back to Top |
|||||||||||||||||||||||||||||||||
Nucleotide Sequence |
Length: 1645 bp Go to nucleotide: FASTA |
|||||||||||||||||||||||||||||||||
Protein Sequence |
Length: 105 bp Go to amino acid: FASTA |
|||||||||||||||||||||||||||||||||
The verified Protein-Protein interaction information |
||||||||||||||||||||||||||||||||||
Other Protein-Protein interaction resources |
String database |
|||||||||||||||||||||||||||||||||
View Microarray data |
Temporarily unavailable |
|||||||||||||||||||||||||||||||||
Comments |
||||||||||||||||||||||||||||||||||