| Tag | Content | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
SG ID |
SG00001962 |
||||||||||
UniProt Accession |
|||||||||||
Theoretical PI |
8.7
|
||||||||||
Molecular Weight |
22265 Da
|
||||||||||
Genbank Nucleotide ID |
|||||||||||
Genbank Protein ID |
|||||||||||
Gene Name |
Gpx4 |
||||||||||
Gene Synonyms/Alias |
|||||||||||
Protein Name |
Glutathione peroxidase |
||||||||||
Protein Synonyms/Alias |
|||||||||||
Organism |
Rattus norvegicus (Rat) |
||||||||||
NCBI Taxonomy ID |
10116 |
||||||||||
Chromosome Location |
|
||||||||||
Function in Stage |
|||||||||||
Function in Cell Type |
|||||||||||
Description |
Temporarily unavailable |
||||||||||
The information of related literatures |
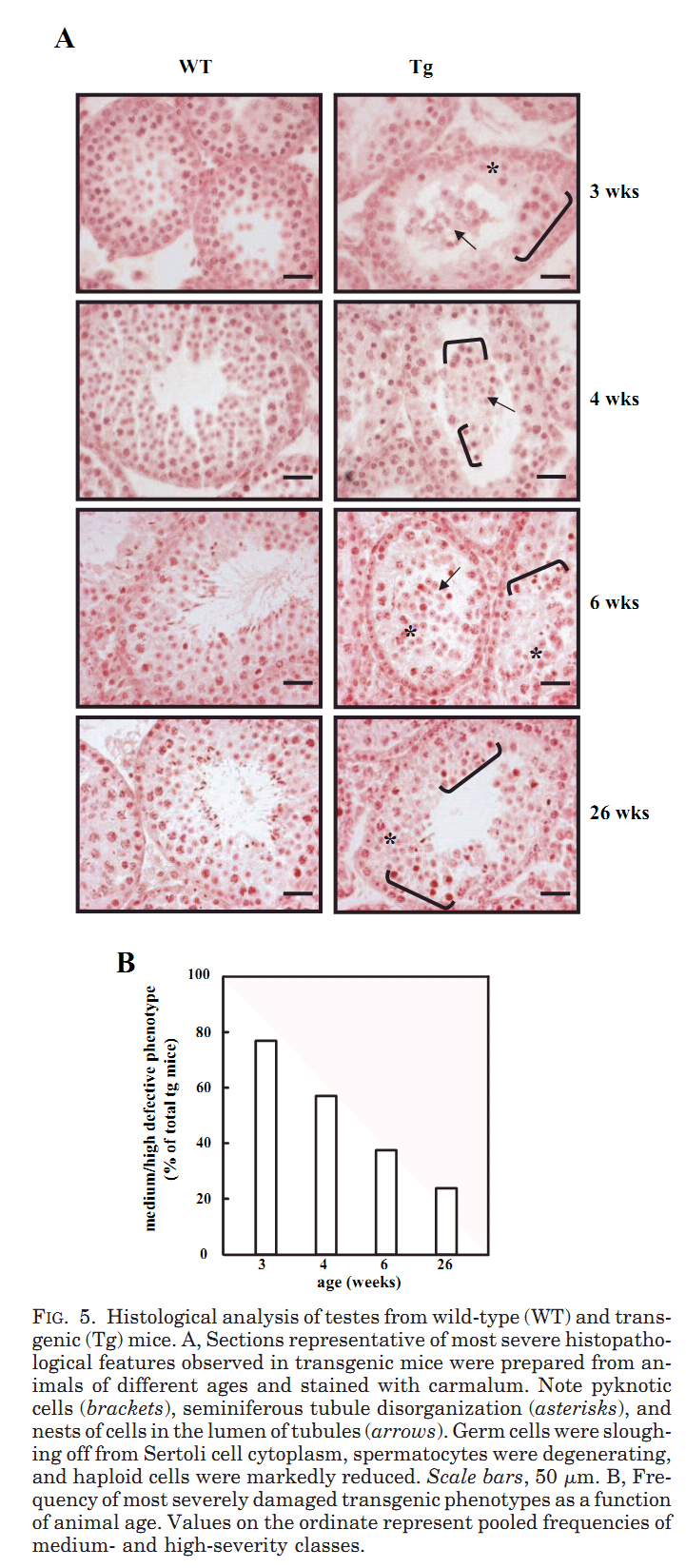
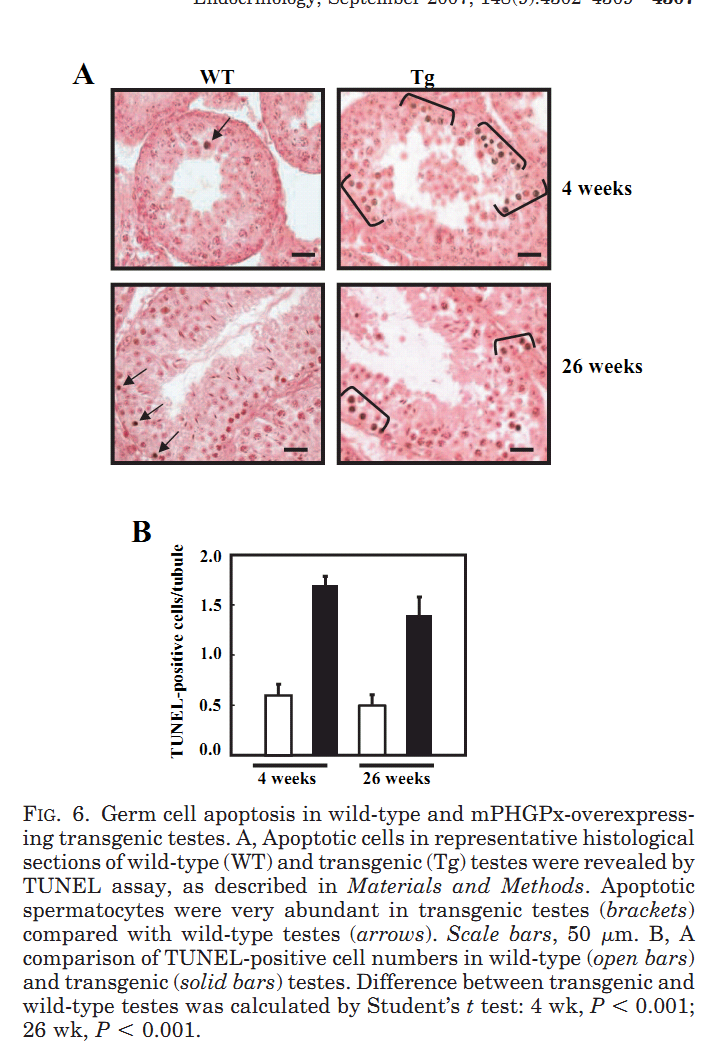
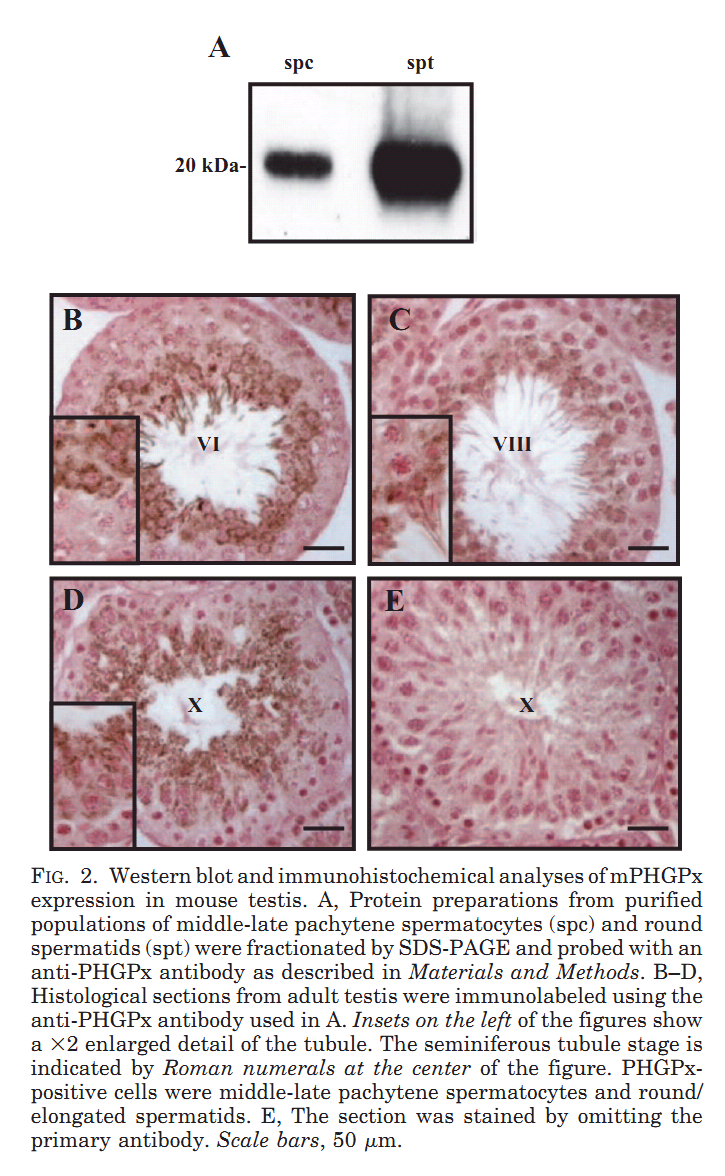
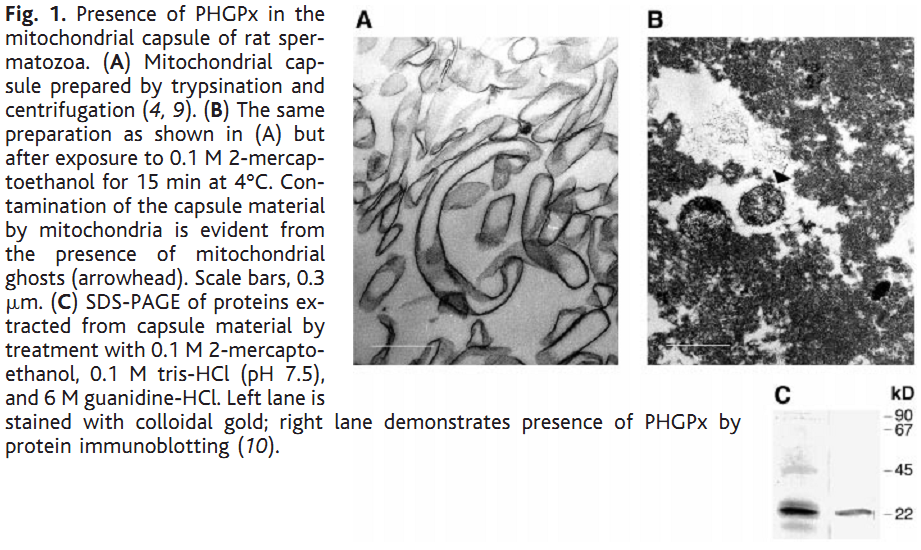
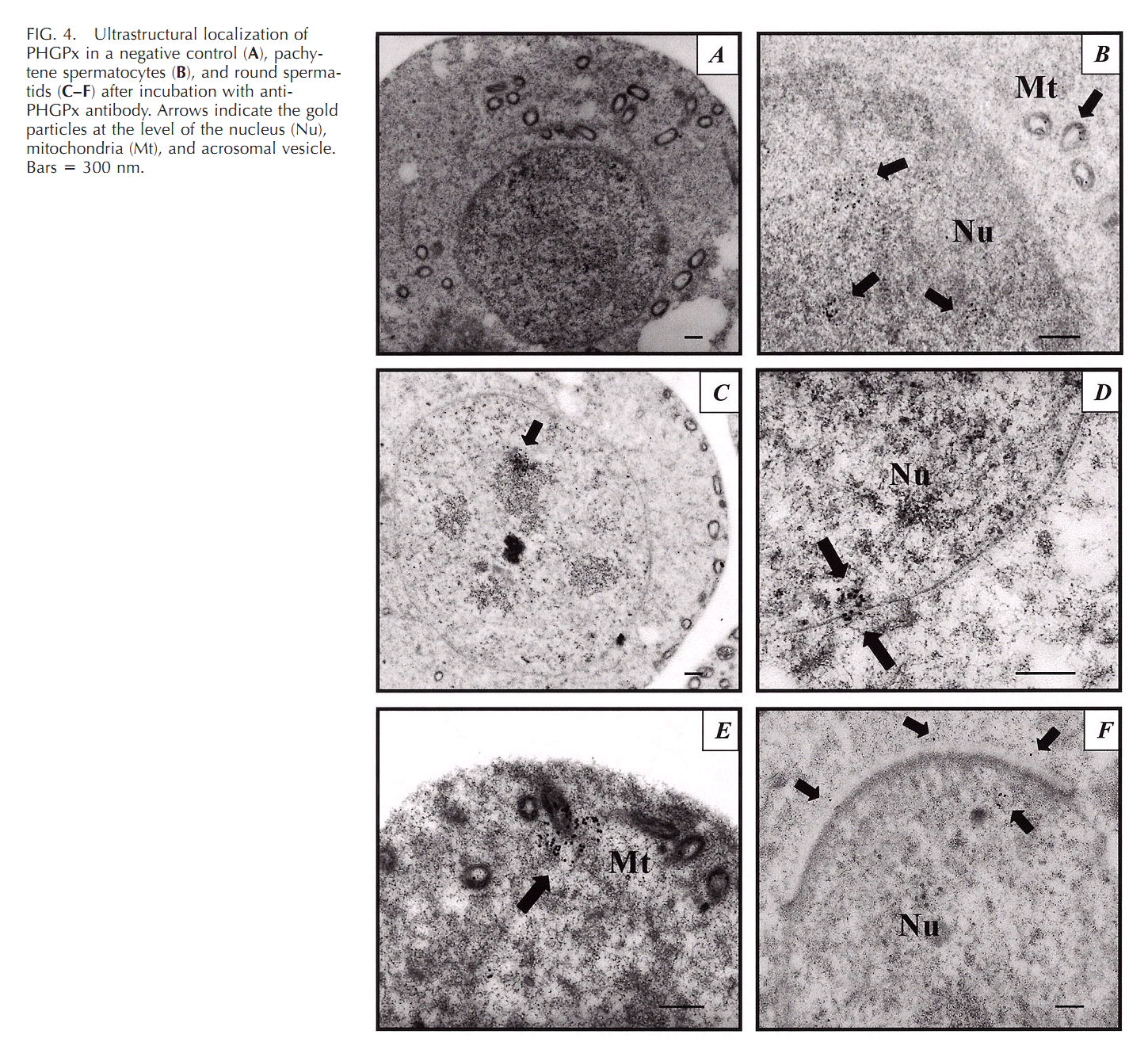
1. R. Puglisi, A. Bevilacqua, G. Carlomagno, A. Lenzi, L. Gandini, M. Stefanini, F. Mangia and C. Boitani (2007) Mice overexpressing the mitochondrial phospholipid hydroperoxide glutathione peroxidase in male germ cells show abnormal spermatogenesis and reduced fertility. Endocrinology 148(9): 4302-9. Abstract To investigate the physiological effects of mitochondrial phospholipid hydroperoxide glutathione peroxidase (mPHGPx) overexpression during early male germ cell differentiation, we have generated transgenic mice bearing the rat mPhgpx coding sequence driven by the mouse synaptonemal complex protein 1 promoter, allowing the transgene to be specifically activated in the testis from the zygotene to diplotene stages of the first meiotic division. Northern/Western blotting and immunocytochemical analyses of endogenous mPHGPx expression during spermatogenesis showed a low enzyme level in middle-late pachytene spermatocytes, but not in earlier meiotic stages, and a significant increase in mPHGPx content in round spermatids. The histological and terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling analysis of transgenic testes revealed a number of spermatogenetic defects, including primary spermatocyte apoptosis, haploid cell loss, and seminiferous epithelium disorganization. In line with these features, adult transgenic male mice also displayed a reduction in fertility. Results obtained in this study suggest that mPHGPx expression is tightly regulated in pachytene spermatocytes, with any spatial-temporal increase in mPHGPx expression resulting in damage to spermatogenesis and eventual loss of haploid cells. Present findings in the mouse may be of interest to human male fertility. PMID: [17540721] 2. F. Ursini, S. Heim, M. Kiess, M. Maiorino, A. Roveri, J. Wissing and L. Flohe (1999) Dual function of the selenoprotein PHGPx during sperm maturation. Science 285(5432): 1393-6. Abstract The selenoprotein phospholipid hydroperoxide glutathione peroxidase (PHGPx) changes its physical characteristics and biological functions during sperm maturation. PHGPx exists as a soluble peroxidase in spermatids but persists in mature spermatozoa as an enzymatically inactive, oxidatively cross-linked, insoluble protein. In the midpiece of mature spermatozoa, PHGPx protein represents at least 50 percent of the capsule material that embeds the helix of mitochondria. The role of PHGPx as a structural protein may explain the mechanical instability of the mitochondrial midpiece that is observed in selenium deficiency. PMID: [10464096] 3. R. Puglisi, F. Tramer, E. Panfili, F. Micali, G. Sandri and C. Boitani (2003) Differential splicing of the phospholipid hydroperoxide glutathione peroxidase gene in diploid and haploid male germ cells in the rat. Biol Reprod 68(2): 405-11. Abstract Phospholipid hydroperoxide glutathione peroxidase (PHGPx, 20 kDa) and sperm nuclei glutathione peroxidase (snGPx, 34 kDa) are two selenoproteins present in mammalian testis and epididymal spermatozoa. They originate from the differential splicing of the PHGPx gene and appear to play important roles in sperm physiology. To determine the stages of spermatogenesis in which they are present, we compared the expression pattern of these two enzymes in highly purified populations of germ cells during specific phases of differentiation. In Northern and Western blotting experiments, both PHGPx transcript and protein were markedly expressed in pachytene spermatocytes and round spermatids. In contrast, the testis-specific snGPx was detected at both the mRNA and protein level only in haploid round spermatids. Accordingly, the developmental analysis of testicular RNAs from rats of different ages first revealed the appearance of PHGPx and snGPx transcripts at Day 20 and Day 30, respectively. Furthermore, both meiotic and postmeiotic cells contained catalytically active PHGPx/snGPx, with higher activity in the haploid cells. The intracellular distribution of PHGPx in mitochondria and nuclei of meiotic cells was demonstrated by immunocytochemical electron microscopy and Western blotting. These findings provide evidence that the PHGPx gene is differentially spliced during the meiotic prophase and haploid cell phases of spermatogenesis. PMID: [12533403] Back to Top |
||||||||||
Figures for illustrating the function of this protein/gene |
|
||||||||||
Function |
|||||||||||
Subcellular Location |
|||||||||||
Tissue Specificity |
|||||||||||
Gene Ontology |
|
||||||||||
Interpro |
|||||||||||
Pfam |
|||||||||||
SMART |
|||||||||||
PROSITE |
PS00460; GLUTATHIONE_PEROXID_1; 1. PS00763; GLUTATHIONE_PEROXID_2; 1. PS51355; GLUTATHIONE_PEROXID_3; 1. Back to Top |
||||||||||
PRINTS |
|||||||||||
Created Date |
18-Oct-2012 |
||||||||||
Record Type |
Experiment identified |
||||||||||
Protein sequence Annotation |
|||||||||||
Nucleotide Sequence |
Length: bp Go to nucleotide: FASTA |
||||||||||
Protein Sequence |
Length: 198 bp Go to amino acid: FASTA |
||||||||||
The verified Protein-Protein interaction information |
| ||||||||||
Other Protein-Protein interaction resources |
String database |
||||||||||
View Microarray data |
Temporarily unavailable |
||||||||||
Comments |
|||||||||||