| Tag | Content | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
SG ID |
SG00001977 |
|||||||||||||||||||||
UniProt Accession |
||||||||||||||||||||||
Theoretical PI |
5.18
|
|||||||||||||||||||||
Molecular Weight |
28055 Da
|
|||||||||||||||||||||
Genbank Nucleotide ID |
||||||||||||||||||||||
Genbank Protein ID |
||||||||||||||||||||||
Gene Name |
Aqp8 |
|||||||||||||||||||||
Gene Synonyms/Alias |
||||||||||||||||||||||
Protein Name |
Aquaporin-8 |
|||||||||||||||||||||
Protein Synonyms/Alias |
AQP-8 |
|||||||||||||||||||||
Organism |
Rattus norvegicus (Rat) |
|||||||||||||||||||||
NCBI Taxonomy ID |
10116 |
|||||||||||||||||||||
Chromosome Location |
|
|||||||||||||||||||||
Function in Stage |
||||||||||||||||||||||
Function in Cell Type |
||||||||||||||||||||||
Description |
Temporarily unavailable |
|||||||||||||||||||||
The information of related literatures |
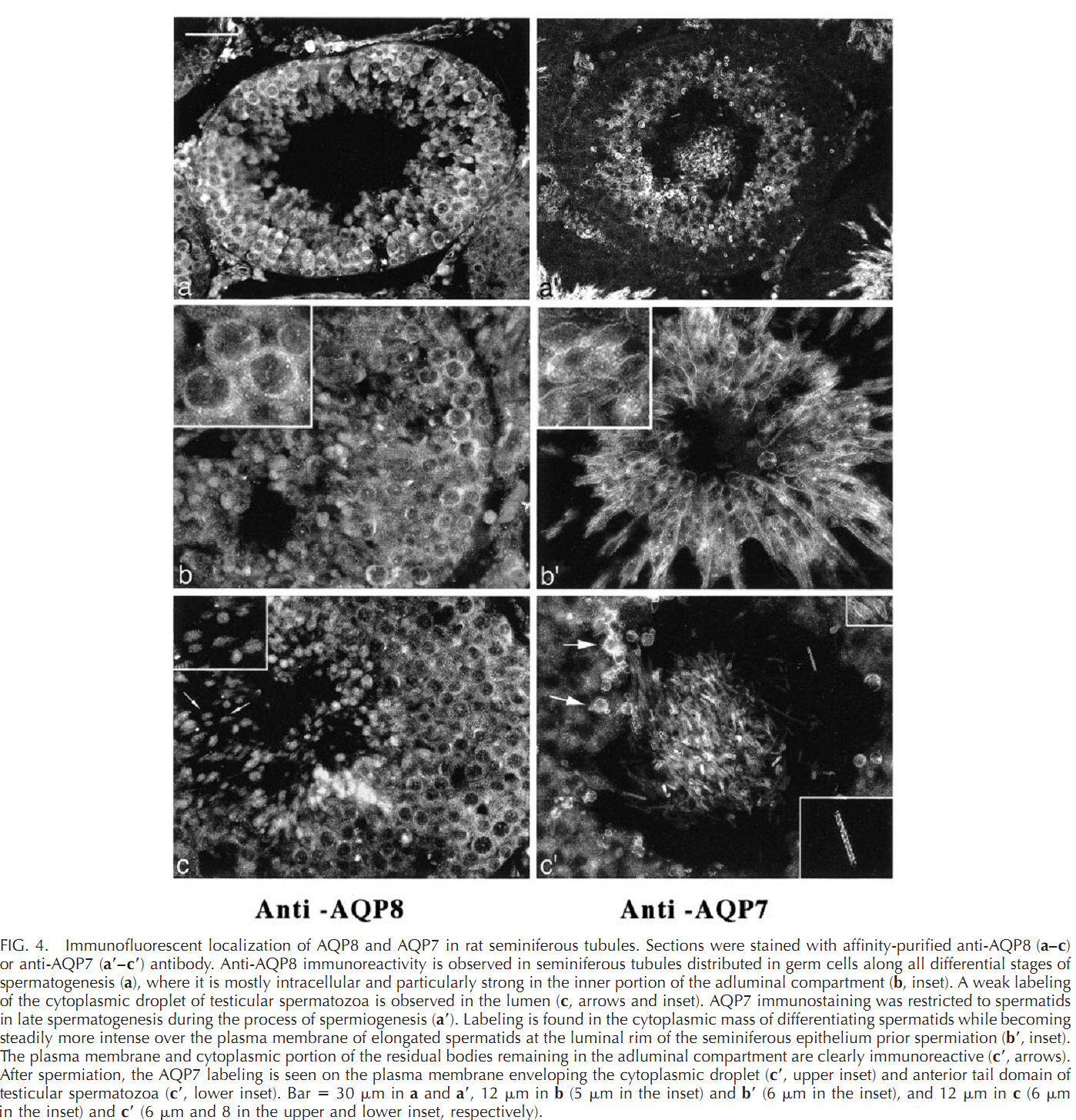
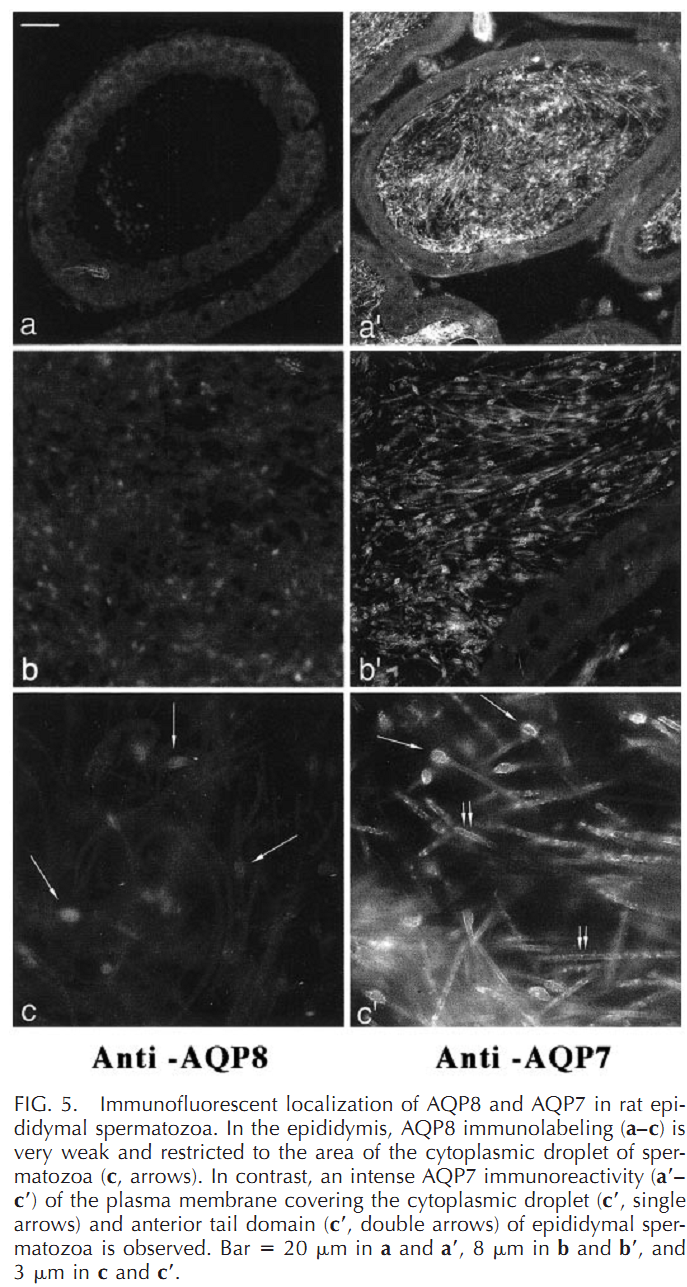
1. G. Calamita, A. Mazzone, Y. S. Cho, G. Valenti and M. Svelto (2001) Expression and localization of the aquaporin-8 water channel in rat testis. Biol Reprod 64(6): 1660-6. Abstract Spermatogenesis and sperm maturation and storage are accompanied by significant movements of water, and multiple aquaporin transmembrane water channels (AQPs) have been recognized in the male reproductive tract. Nevertheless, the involvement of aquaporins in male reproductive physiology is mostly unknown. Here the expression and localization of AQP8 in rat spermatogenesis is defined and compared to that of AQP7, another aquaporin expressed in male germ cells. AQP8 mRNA was found in testis but not in epididymis, whereas the AQP7 transcript was present in both locations. By immunoblotting, the AQP8 protein was detected as a 25-kDa band and a 32- to 40-kDa diffuse component corresponding to the core and glycosylated protein, respectively. Membrane fractionation revealed AQP8 both in microsomal and plasma membrane-enriched fractions of rat testis while no apparent bands were detected in epididymis. AQP7 appeared as a 23- to 24-kDa band and was found both in testis and epididymis. By immunofluorescence, AQP8 labeling was found intracellularly as well as over the plasma membrane of germ cells throughout spermatogenesis. AQP7 was present in spermatids and spermatozoa and was predominant over the plasma membrane. AQP8 may be involved in the cytoplasmic condensation occurring during differentiation of spermatids into spermatozoa and in the generation of seminiferous tubule fluid. PMID: [11369592] 2. G. Calamita, A. Mazzone, A. Bizzoca and M. Svelto (2001) Possible involvement of aquaporin-7 and -8 in rat testis development and spermatogenesis. Biochem Biophys Res Commun 288(3): 619-25. Abstract Fluid secretion and reabsorption are of central importance in male reproductive (MR) physiology. However, the related molecular mechanisms are poorly known. Here, potential roles for AQP7 and AQP8, two aquaporin water channels abundantly expressed in the MR tract, were investigated by studying their expression and distribution in the developing testis of the Wistar rat. By semiquantitative RT-PCR and immunoblotting, first expression of AQP7 was noted at postnatal day 45 (P45), with levels increasing substantially at P90 and remaining at high levels thereafter. AQP8 began to be expressed at P15, rapidly increased until P20, and remained fairly stable thereafter. Immunohistochemical analyses demonstrated AQP7 in elongated spermatids, testicular spermatozoa, and residual bodies at P45 with increased signal intensity thereafter. AQP8 was observed in primary spermatocytes from P20 to P30 and, in elongated spermatids, residual bodies and Sertoli cells at P30 and thereafter. The ontogeny and distribution of AQP7 and AQP8 in rat testis suggest involvement in major physiologic changes in testis development and spermatogenesis. PMID: [11676488] 3. Y. Kageyama, K. Ishibashi, T. Hayashi, G. Xia, S. Sasaki and K. Kihara (2001) Expression of aquaporins 7 and 8 in the developing rat testis. Andrologia 33(3): 165-9. Abstract Aquaporin (AQP) is a molecule that facilitates water permeability through cell membranes and is widely distributed in the mammalian body. Among members of the AQP family, AQP7 and AQP8 are predominantly expressed in the testis. This paper examined the expression of these two AQPs in the developing rat testis, using northern blot and immunohistochemistry to visualize their roles during the progression of spermatogenesis in the developing testis. In adult rats, expression of AQP7 was localized to spermatids, whereas AQP8 protein was observed in spermatocytes. In the developing testis, transcripts of AQP7 became detectable between 23 and 25 days post-partum, when round spermatids have been reported to appear. On the other hand, AQP8 mRNA was first detected between 13 and 16 days post-partum, consistent with the reported first appearance of spermatocytes (13-14 days). AQP7 and AQP8 proteins were detectable at 28 days post-partum. The results suggest that AQP7 and AQP8 appear in the testis in accordance with the maturation of germ cells, and are involved in spermatogenesis. PMID: [11380332] Back to Top |
|||||||||||||||||||||
Figures for illustrating the function of this protein/gene |
|
|||||||||||||||||||||
Function |
Forms a water-specific channel; mercury-sensitive. Itmay have an important role in spermatogenesis, in fertilization,and in the secretion of pancreatic juice and saliva. Back to Top |
|||||||||||||||||||||
Subcellular Location |
Membrane; Multi-pass membrane protein. |
|||||||||||||||||||||
Tissue Specificity |
Highly expressed in sperm, pancreas and liver.Some expression has been found in salivary gland and absorptivecolonic epithelial cells. |
|||||||||||||||||||||
Gene Ontology |
|
|||||||||||||||||||||
Interpro |
||||||||||||||||||||||
Pfam |
||||||||||||||||||||||
SMART |
||||||||||||||||||||||
PROSITE |
||||||||||||||||||||||
PRINTS |
||||||||||||||||||||||
Created Date |
18-Oct-2012 |
|||||||||||||||||||||
Record Type |
Experiment identified |
|||||||||||||||||||||
Protein sequence Annotation |
CHAIN 1 263 Aquaporin-8. /FTId=PRO_0000063963. TOPO_DOM 1 38 Cytoplasmic (Potential). TRANSMEM 39 59 Helical; (Potential). TOPO_DOM 60 86 Extracellular (Potential). TRANSMEM 87 107 Helical; (Potential). TOPO_DOM 108 109 Cytoplasmic (Potential). TRANSMEM 110 130 Helical; (Potential). TOPO_DOM 131 158 Extracellular (Potential). TRANSMEM 159 179 Helical; (Potential). TOPO_DOM 180 185 Cytoplasmic (Potential). TRANSMEM 186 206 Helical; (Potential). TOPO_DOM 207 230 Extracellular (Potential). TRANSMEM 231 251 Helical; (Potential). TOPO_DOM 252 263 Cytoplasmic (Potential). MOTIF 94 96 NPA 1. MOTIF 212 214 NPA 2. CARBOHYD 141 141 N-linked (GlcNAc...) (Potential). Back to Top |
|||||||||||||||||||||
Nucleotide Sequence |
Length: 1463 bp Go to nucleotide: FASTA |
|||||||||||||||||||||
Protein Sequence |
Length: 263 bp Go to amino acid: FASTA |
|||||||||||||||||||||
The verified Protein-Protein interaction information |
| |||||||||||||||||||||
Other Protein-Protein interaction resources |
String database |
|||||||||||||||||||||
View Microarray data |
Temporarily unavailable |
|||||||||||||||||||||
Comments |
||||||||||||||||||||||