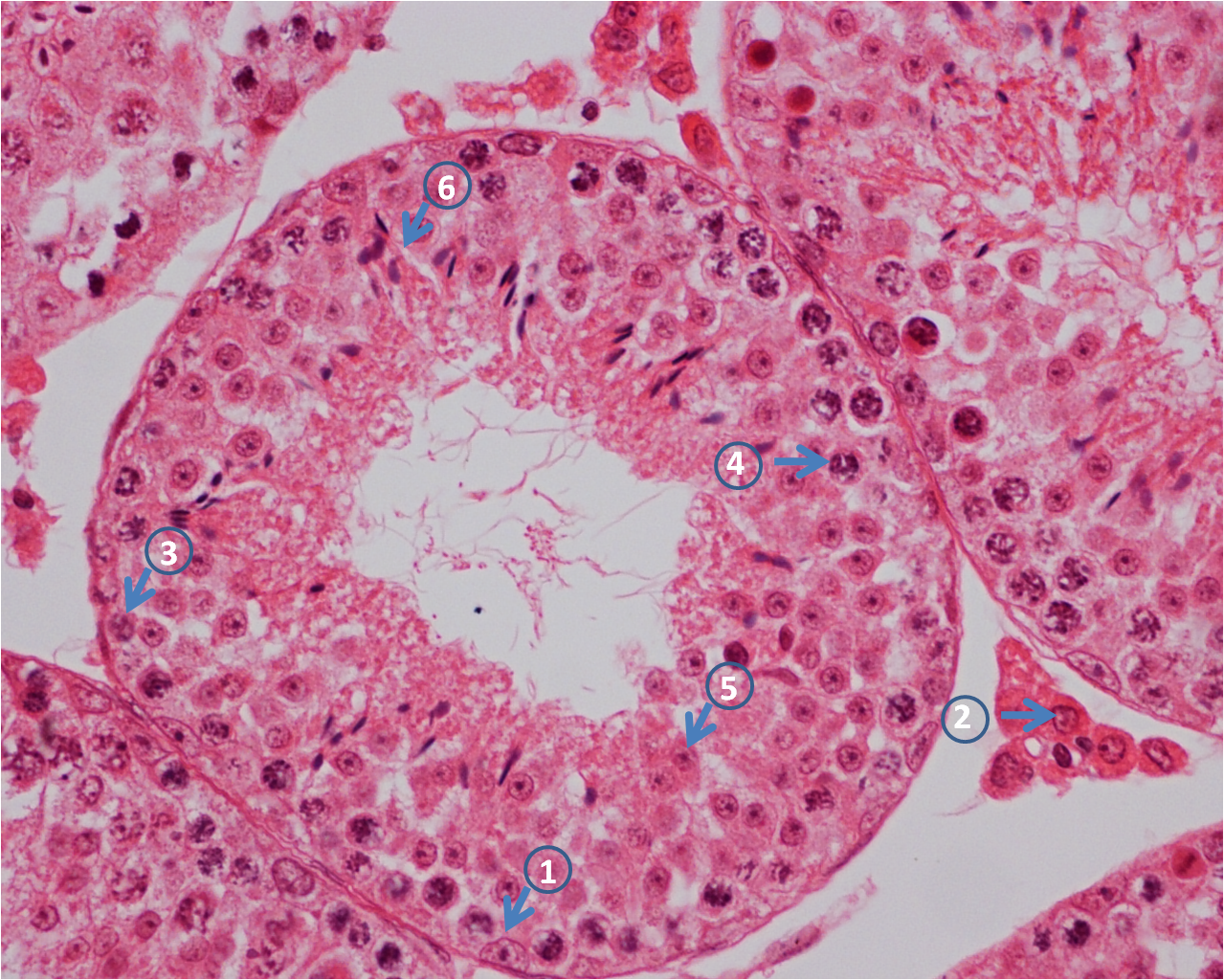
Introduction
Spermatogenesis is a sophisticated biological process responsible for development of spermatozoa from spermatogonial stem cells (1, 2, 3, 4). The process includes stem-cell renewal and differentiation, genome reorganization and half reduction, and genome repackaging and cellular transformation (5, 6). During the continuous process, the intricate interaction between spermatogenetic cell and somatic cell is elaborately regulated. Generally, it has been divided into three distinct stages: premeiotic, meiotic, and postmeitoic stages (7). At the premeiotic stage, renewal and differentiation of spermatogonial stem cells (SSC) and mitotic proliferation of spermatogonia occur (2, 8, 9). After that, the type B spermatogonia enter into meiosis, and are transformed into spermatocytes (8). During the meiotic stage, the genomes of primary spermatocytes are halved into secondary spermatocytes; In the process of postmeiotic stage (also termed as spermiogenesis), the secondary spermatocytes are formed into round spermatids because of chromatin and nuclear condensation (10). Transformation of round spermatids into enlongated spermatids, finally spermatozoa, ascribes to rearrangement of cytoskeletal structure and genome repackaging (11, 12). In the whole stages, two somatic cells play a vital role in mammalian spermatogenesis, one of them is leydig cells, which secretes testosterone, stimulating sertoli cells (the other one of them),direct contact with differentiating germ cells, to support spermatogenesis in the seminiferous tubules(see figure below) (5, 13, 14, 15, 16, 17). Inappropriate coordination of these events result in spermatogenic pathologies such as spermatogenic arrest, Sertoli cell only syndrome (SCO) and even sterility. Although great progress has been made by the genetic modification of model animals, the molecular mechanisms of spermatogenesis still remain to be discovered.
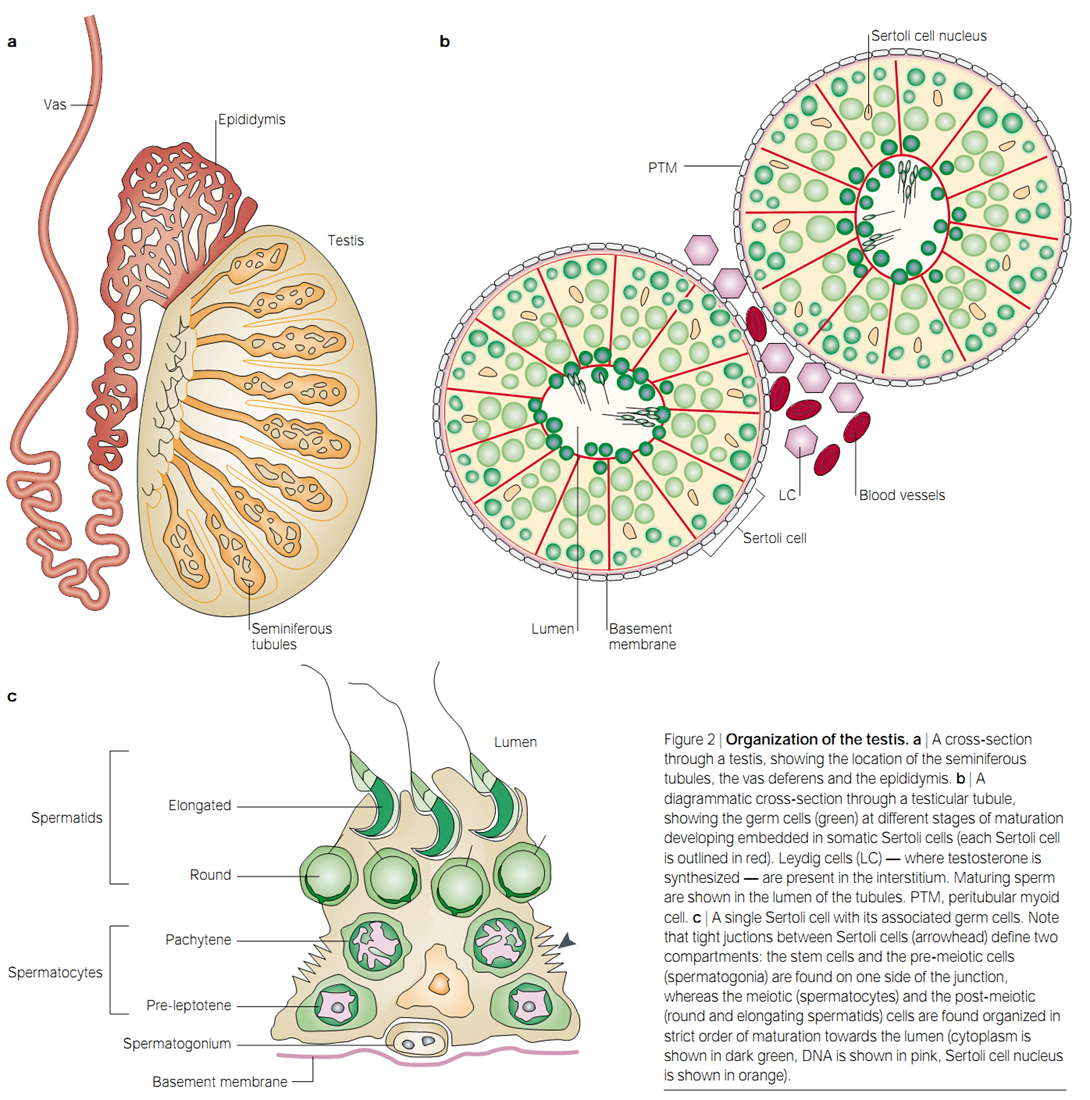
The spermatogenesis is long lasting and complex process, although many proteins have been identified to be implicated in it, an integrated resource in this area still remains unavailable. In this work, we collected all proteins identified to play roles or potential roles in spermatogenesis. After manually curated, XX records correspondent to XX proteins has been confirmed to be involved in the spermatogenesis. Then, an integrated and searchable database Spermtogenesis Online 1.0 has been founded. It is the first integrative resource embracing the most of identified components and related scientific information of spermatogenesis.
Currently, the SpermatogenesisOnline 1.0 database has been updated on Jun 13, 2012, containing 22901 unique protein entries. The online service of SpermatogenesisOnline 1.0 was implemented in PHP + MySQL + JavaScript. The database will be updated routinely as new Spermatogenesis genes/proteins are reported.
References for Introduction Click to open
Click to close
1. de Rooij, D.G. (1998). Stem cells in the testis. Int J Exp Pathol 79, 67-80. PMID: 9709376
2. de Rooij, D.G., and Grootegoed, J.A. (1998). Spermatogonial stem cells. Curr Opin Cell Biol 10, 694-701. PMID: 9914171
3. Huckins, C. (1971). The spermatogonial stem cell population in adult rats. I. Their morphology, proliferation and maturation. Anat Rec 169, 533-557. PMID: 5550532
4. Oakberg, E.F. (1971). Spermatogonial stem-cell renewal in the mouse. Anat Rec 169, 515-531. PMID: 5550531
5. Cooke, H.J., and Saunders, P.T. (2002). Mouse models of male infertility. Nat Rev Genet 3, 790-801. PMID: 12360237
6. Guan, K., Nayernia, K., Maier, L.S., Wagner, S., Dressel, R., Lee, J.H., Nolte, J., Wolf, F., Li, M., Engel, W., et al. (2006). Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 440, 1199-1203. PMID: 16565704
7. Holstein, A.F., Schulze, W., and Davidoff, M. (2003). Understanding spermatogenesis is a prerequisite for treatment. Reprod Biol Endocrinol 1, 107. PMID: 14617369
8. Clermont, Y. (1962). Quantitative analysis of spermatogenesis of the rat: a revised model for the renewal of spermatogonia. Am J Anat 111, 111-129. PMID: 13879927
9. de Rooij, D.G., and Russell, L.D. (2000). All you wanted to know about spermatogonia but were afraid to ask. J Androl 21, 776-798. PMID: 11105904
10. Meistrich, M. L. (1989) in Histone and Basic Nuclear Protein Transitions in Mammalian Spermatogenesis, eds. Hnilica, G., Stein, G. & Stein, J. (CRC,Orlando, FL), pp. 165-182.
11. Clermont Y, Oko R,Hermo L. 1993. Cell biology of mammalian spermiogenesis. In Cell and Molecular Biology of the Testis, ed. C Desjardins, LL Ewing, 1:332–76. New York: Oxford Univ. Press.
12. Balhorn, R., Weston, S., Thomas, C., and Wyrobek, A.J. (1984). DNA packaging in mouse spermatids. Synthesis of protamine variants and four transition proteins. Exp Cell Res 150, 298-308. PMID: 6692853
13. Bremner, W.J., Millar, M.R., Sharpe, R.M., and Saunders, P.T. (1994). Immunohistochemical localization of androgen receptors in the rat testis: evidence for stage-dependent expression and regulation by androgens. Endocrinology 135, 1227-1234. PMID: 8070367
14. Connell, C.J. (1976). A scanning electron microscope study of the interstitial tissue of the canine testis. Anat Rec 185, 389-401. PMID: 970658
15. Griswold, M.D. (1998). The central role of Sertoli cells in spermatogenesis. Semin Cell Dev Biol 9, 411-416. PMID: 9813187
16. Prince, F.P. (2001). The triphasic nature of Leydig cell development in humans, and comments on nomenclature. J Endocrinol 168, 213-216. PMID: 11182757
17. Zhu, L.J., Hardy, M.P., Inigo, I.V., Huhtaniemi, I., Bardin, C.W., and Moo-Young, A.J. (2000). Effects of androgen on androgen receptor expression in rat testicular and epididymal cells: a quantitative immunohistochemical study. Biol Reprod 63, 368-376. PMID: 10906039
Citation
Zhang, Y.#, L. Zhong#, B. Xu#, Y. Yang#, R. Ban, J. Zhu, H. J. Cooke, Q. Hao and Q. Shi* (2013). SpermatogenesisOnline 1.0: a resource for spermatogenesis based on manual literature curation and genome-wide data mining. Nucleic Acids Res 41(Database issue): D1055-1062.
Keywords
1. Sertoli cell 2. Leydig cell 3. Spermatogonium 4. Spermatocyte 5. Spermatid 6. Spermatozoon